Project Design
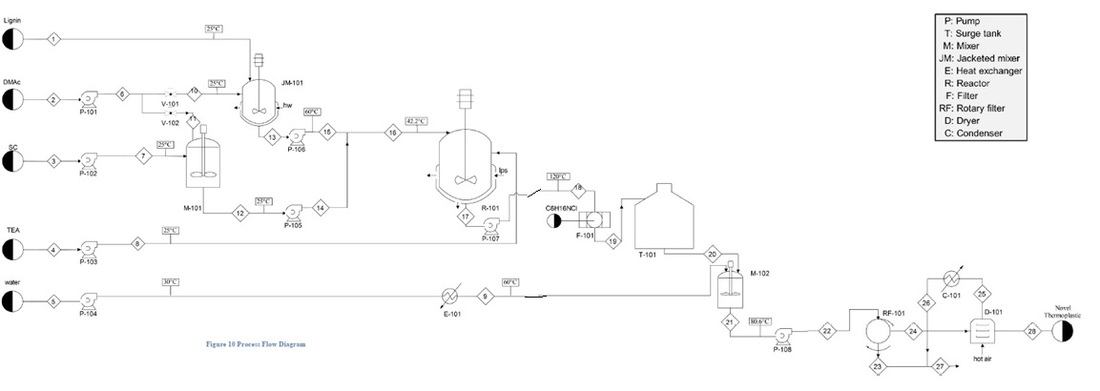
Process Flow Diagram
Lignin powder, Sebacoyl chloride (C10H16O2Cl2), Liquid dissolver N,N-dimethylacetamide (DMAc), and catalyst Triethylamine (TEA) are the raw materials for the reaction. First, lignin as powder is transferred to a jacket-heated mixer (Figure 6), where it is dissolved in N,N-dimethylacetamide (DMAc) and mixed at 60ºC for 1 hr. Hot oil / water can be used as the heating medium. The solution stream is ready and called first solution. Furthermore, this mixer can provide heat to the blended materials by injecting hot medium in the jacket that is included on the outside of the mixer cylinder. A second solution is prepared by dissolving Sebacoyl chloride (C10H16O2Cl2) (SC) in N,N-dimethylacetamide (DMAc), and it will take ½ hr to be ready at normal condition of room temperature. After that, the two solutions prepared above will be fed to a semi-batch jacketed reactor where the reaction takes place at 120oC for 10 hrs. This part of the process considered as the heart of the process. The HCl produced as a by-product prevents the polymerization reaction, by acting as an inhibitor. The triethylamine (TEA) used here to eliminate HCl formation and let the reaction proceed by forming triethylamine hydrochloride (C6H16NCl) as a precipitate. We consider that the TEA has another important factor in determining the molecular weight or chain configuration of the lignin co-polyester because it alters the polymerization rate as well as the termination step. Kraft lignin was polymerized with sebacoyl chloride (SC) in poly-esterification reaction in the presence of triethylamine (TEA) in N,N-dimethylacetamide (DMAc),and the molecular weight of the synthesized polymer is controlled by the polymerization temperatures and ratio of lignin macromonomers, [COCl]/[OH]. The product from the reactor (third solution) will be the synthesised thermoplastic co-polyester solution. The 120ºC and [COCl]/[OH] molar ratio = 2 have been chosen here because they give the highest molecular weight (Mw = 40,000 g/mol), molecule number (Mn = 13,000) and the highest molecular weight distribution (MWD = Mw/Mn = 3.0). After that, the third solution stream passed through a filtration process where the impurities are removed as a precipitate. The filtrate stream then stored in surge tank to complete the process as a continuous process in order to use smaller volume of the coming equipment. Previous filtrate dissolved in water and allowed to form a suspension in the solution. Moreover, this solution is then fed to a rotary vacuum filter where the desired solid product (cake) adheres to the outside of the drum, which then passes a knife, cutting off the solids and a small portion of the filter media to reveal a fresh media surface that will enter the liquid as the drum rotates. The knife advances automatically as the surface is removed (cake). Finally, this cake product is sent to a drier where it is exposed to air with low relative humidity (RH) which will lead to evaporation of the moisture in the grain cake and the desired product is obtained (thermoplastic copolymer).